What are Grignard Reagents?
A Grignard reagent has a formula RMgX where X is a halogen, and R is an alkyl or aryl (based on a benzene ring) group. For the purposes of this page, we shall take R to be an alkyl group.
A typical Grignard reagent might be CH3CH2MgBr.
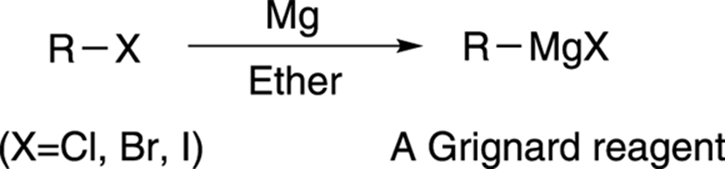
The Preparation of a Grignard Reagent:
Grignard reagents are made by adding the halogenoalkane to small bits of magnesium in a flask containing ethoxyethane (commonly called diethyl ether or just “ether”). The flask is fitted with a reflux condenser, and the mixture is warmed over a water bath for 20 – 30 minutes.
![]()
Everything must be perfectly dry because Grignard reagents react with water (see below).
Any reactions using the Grignard reagent are carried out with the mixture produced from this reaction. You can’t separate it out in any way.
Reactions of Grignard Reagents:
Grignard Reagents and Water:
Grignard reagents react with water to produce alkanes. This is the reason that everything has to be very dry during the preparation above.
For example:
![]()
The inorganic product, Mg(OH)Br, is referred to as a “basic bromide”. You can think of it as a sort of half-way stage between magnesium bromide and magnesium hydroxide.
Grignard Reagents and Carbon Dioxide:
Grignard reagents react with carbon dioxide in two stages. In the first, you get an addition of the Grignard reagent to the carbon dioxide.
Dry carbon dioxide is bubbled through a solution of the Grignard reagent in ethoxyethane, made as described above.
For example:
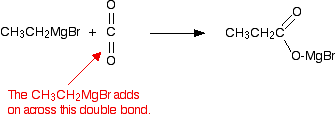
The product is then hydrolysed (reacted with water) in the presence of a dilute acid. Typically, you would add dilute sulphuric acid or dilute hydrochloric acid to the solution formed by the reaction with the CO2.
A carboxylic acid is produced with one more carbon than the original Grignard reagent.
The usually quoted equation is (without the red bits):
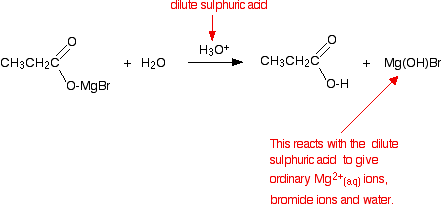
Almost all sources quote the formation of a basic halide such as Mg(OH)Br as the other product of the reaction. That’s actually misleading because these compounds react with dilute acids. What you end up with would be a mixture of ordinary hydrated magnesium ions, halide ions and sulphate or chloride ions – depending on which dilute acid you added.

These notes are good
Thanks