Principles of Ion Exchange to Soften Water:
Calcium and magnesium ions are atoms having a positive electrical charge, as do sodium and potassium ions. Ions of the same charge can be exchanged. In the ion exchange process, a granular substance (usually a resin) that is coated with sodium or potassium ions comes into contact with water containing calcium and magnesium ions.
Two positively charged sodium or potassium ions are exchanged (released into the water) for every calcium or magnesium ion that is held by the resin. This “exchange or trade” happens because sodium or potassium are loosely held by the resin. In this way, calcium and magnesium ions responsible for hardness are removed from the water, held by the resin, and replaced by sodium or potassium ions in the water.
This process makes water “soft.” Eventually, a point is reached when very few sodium or potassium ions remain on the resin, thus no more calcium or magnesium ions can be removed from the incoming water. The resin at this point is said to be “exhausted” or “spent,” and must be “recharged” or “regenerated.
Ion Exchange or De-Ionization or De-Mineralization Process:
Ion-exchange resins are insoluble, cross-linked, long chain organic polymers with a microporous structure, and the “functional groups” attached to the chains are responsible for the ion-exchanging properties.
Resins containing acidic functional groups (− C00H, − SO3H etc.) are capable of exchanging their H+ ions with other cations, which comes in their contact; whereas those containing basic functional groups (− NH2 = NH as hydrochloride salt) are capable of exchanging their anions with other anions, which comes in their contact. The ion-exchange resins may be classified as:
(i) Cation exchange resins (RH+) are mainly styrene-divinyl benzene copolymers, which on sulphonation or carboxylation, become capable to exchange their hydrogen ions with the cations in the water.
(ii) Anion exchange resins (R’OH–) are styrene-divinyl benzene or amine formaldehyde copolymers, which contain amino or’ quaternary ammonium or quaternary phosphonium or tertiary sulphonium groups as an integral part of the resin matrix. These, after treatment with dil. NaOH solution, become capable to exchange their OH– anions with anions in water.
Process of Ion Exchange to Soften Water:
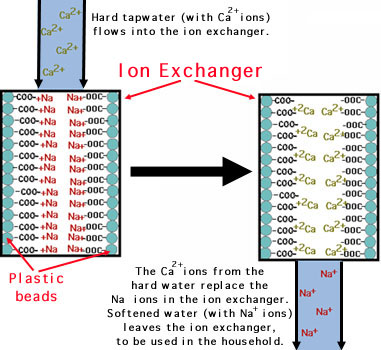
The hard water is passed first through cation exchange column, which removes all the cations like Ca2+, Mg2+, etc. from it, and equivalent amount of H+ ions are released from this column to water. Thus;
2RH+ + Ca2+ ——–.> R2Ca2+ + 2H+
2RH+ + Mg2+ ——.> R2Mg2+ + 2H+
After cation exchange column, the hard water is passed through anion exchange column, which removes all the anions like SO42−, Cl−, etc. present in the water and equivalent amount of OH− ions are released from this column to water. Thus:
R’OH– + Cl– —..——.> R’Cl– + OH–
2R’OH– + SO42- —..–> R’2SO42- + 2OH–
2R’OH– + CO32- —-.-> R’2CO32- + 2OH–
H+ and OH– ions (released from cation exchange and anion exchange columns respectively) get combined to produce water molecule.
H+ + OH- –> H2O
Thus, the water coming out from the exchanger is free from cations as well as anions. Ion-free water, is known as deionized or demineralised water.
Regeneration:
When capacities of cation and anion exchangers to exchange H+ and OH– ions respectively are lost, they are then said to be exhausted. The exhausted cation exchange column is regenerated by passing a solution of dil. HCl or dil H2SO4. The regeneration can be represented as :
R2Ca2+ + 2H+ —-> 2RH+ + Ca2+ (washing)
The column is washed with deionised water and washing (which contains Ca2+, Mg2+, etc. and Cl– or SO42- ions) is passed to sink or drain The exhausted anion exchange column is regenerated by passing a solution of dil. NaOH. The regeneration can be represented as :
R’2S042- + 2OH– —> 2R’OH– + SO42-(washing)
The column is washed with deionised water and washing (which contains Na+ and SO42- or Cl– ions) is passed to silk or drain. The regenerated ion exchange resins are then used again.
Advantages of Ion Exchange Process:
- The process can be used to soften highly acidic or alkaline waters.
- It produces water of very low hardness (say 2 ppm). So it is very good for treating water for use in high-pressure boilers.
Disadvantages of Ion Exchange Process:
- The equipment is costly and more expensive chemicals are needed.
- If water contains turbidity, then the output of the process is reduced. The turbidity must be below 10 ppm. If it is more, it has to be removed first by coagulation and then by filtration.
