What is a Fullerene?
A fullerene, is an allotrope of carbon whose molecule consists of carbon atoms connected by single and double bonds so as to form a closed or partially closed mesh, with fused rings of five to seven atoms. The molecule may be a hollow sphere, ellipsoid, tube, or many other shapes and sizes.
What is the structure of fullerenes?
Fullerenes are molecules of carbon atoms with hollow shapes. Their structures are based on hexagonal rings of carbon atoms joined by covalent bonds. Some fullerenes include rings with five or seven carbon atoms. Two examples of fullerenes are buckminsterfullerene and nanotubes.
Buckminsterfullerene
Buckminsterfullerene was the first fullerene to be discovered. Its molecules are made up of 60 carbon atoms joined together by strong covalent bonds. Molecules of C60 are spherical.
There are weak intermolecular forces between molecules of buckminsterfullerene. These need little energy to overcome, so buckminsterfullerene is slippery and has a low melting point.
Covalent structure of buckminsterfullerene
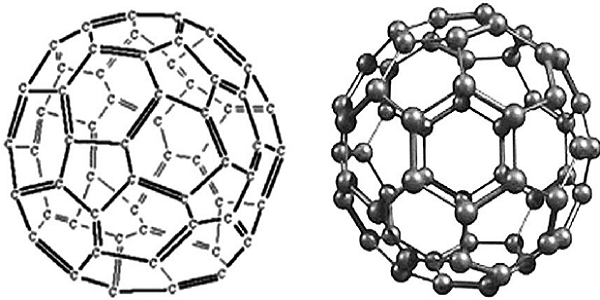
Buckminsterfullerene has sixty carbon atoms joined by covalent bonds
Nanotubes
A nanotube is like a layer of graphene, rolled into a cylinder. The length of a nanotube is very long compared to its width, so nanotubes have high length to diameter ratios.
Nanotubes have high tensile strength, so they are strong in tension and resist being stretched. Like graphene, nanotubes are strong and conduct electricity because they have delocalised electrons.
These properties make nanotubes useful for nanotechnology, electronics and specialised materials.
Covalent structure of Nanotubes
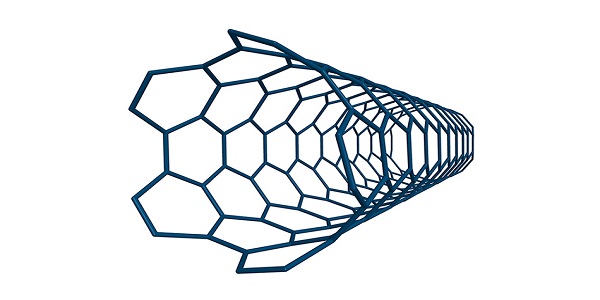
Nanotubes can be several millimetres long but only a few nanometres wide
The discovery of fullerenes greatly expanded the number of known allotropes of carbon, which had previously been limited to graphite, diamond, and amorphous carbon such as soot and charcoal. They have been the subject of intense research, both for their chemistry and for their technological applications, especially in materials science, electronics, and nanotechnology
